
It is always a part of a compound with other elements. As with all reactive elements, bromine is never found alone in nature. For example, we write boron as 1s22s22p1, meaning the n1 0 orbital. Sodium bromide is a compound found in seawater. Thus we label the multi-electron atoms using the notation for hydrogen-atom orbitals. You will find bromine in many salt compounds with alkali metals. Like the other halogens, bromine has seven electrons in its outer shell and is very reactive. Its companions include fluorine, chlorine, and iodine. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here.īromine is a member of the halogen family of elements. 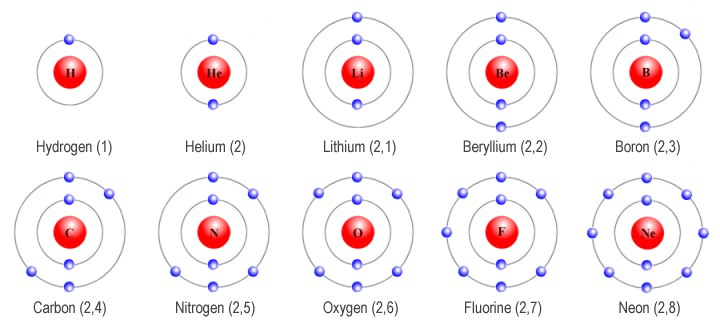
They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. In addition to s and p orbitals, there are two other sets of orbitals which become available for electrons to inhabit at higher energy levels. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. Depending on the choice of 2p orbital and spin function, it is possible to write down. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. The ground state electron configuration of the boron atom is 1s 22s 22p 1. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Now we're working with the fourth period/row in the table of elements. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. Using only the periodic table below, write the expected ground-state electron configurations for a.) the. 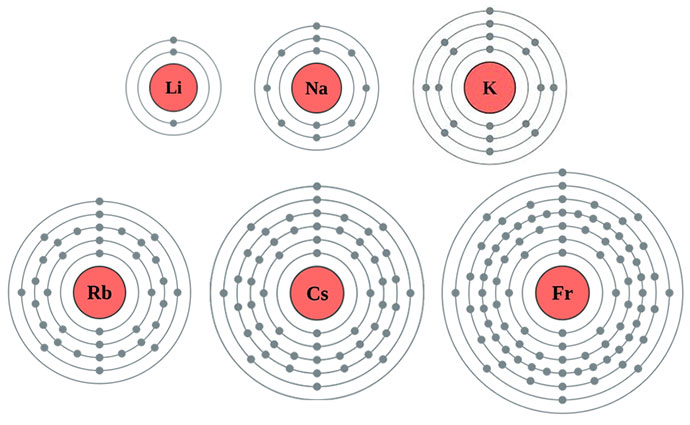
That box on the left has all of the information you need to know about one element. the halogen with electrons in the 6p atomic orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
